AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
C element periodic table12/7/2023 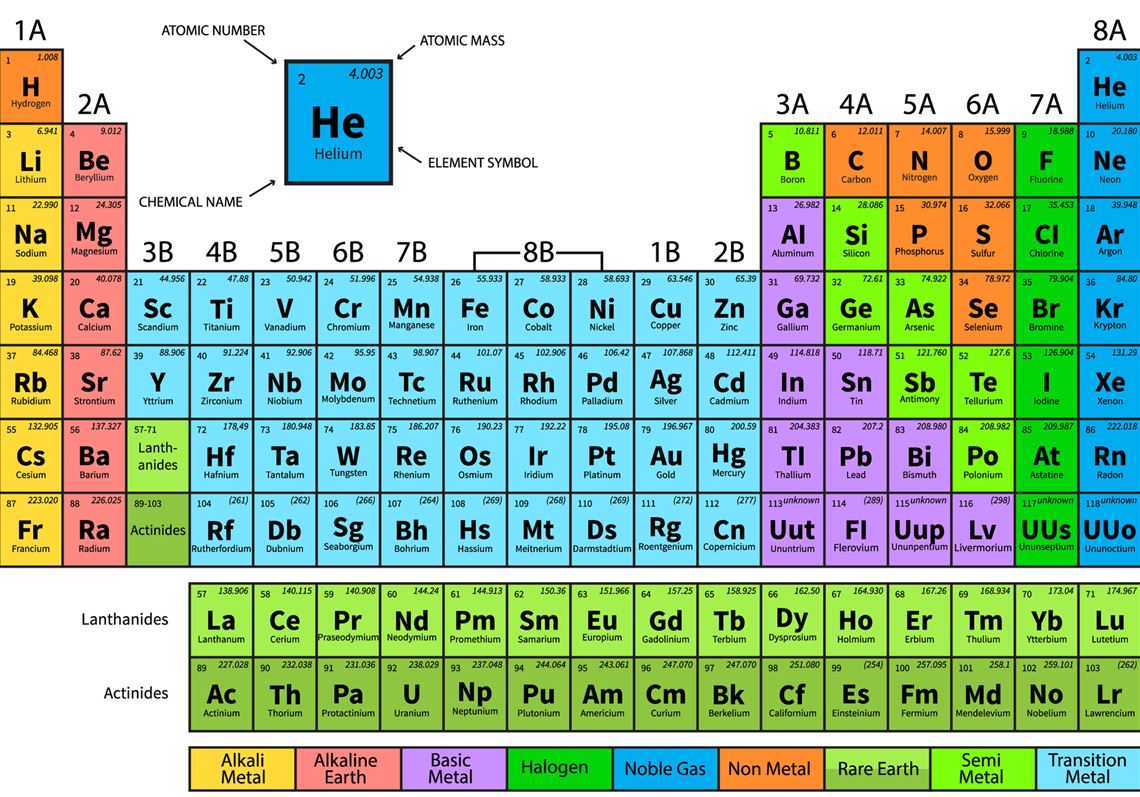
Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. Most window glass is made by floating molten. A niobium-tin alloy is used for superconducting magnets. Alloys of tin are important, such as soft solder, pewter, bronze and phosphor bronze. Please report any accidental mistake in the above statistics on chemical elements.The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. It takes a high polish and is used to coat other metals to prevent corrosion, such as in tin cans, which are made of tin-coated steel. It alloys with copper, tin, and zinc to form amalgams, or liquid alloys. Mercury is silvery white and slowly tarnishes in moist air. Mercury is the only elemental metal that is liquid at room temperature. A chemical element, often simply called an element, is a type of which has the same number of protons in its atomic nucleus (i.e., the same atomic number 1 The definitive visualisation of all 118 elements is the periodic table. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.Ĭlick here: for a schematic overview of the periodic table of elements in chart formĭo you need to know the weight of some molecules? Try our Molecular Weight Calculator! Mercury, chemical element, liquid metal of Group 12 of the periodic table. This is a list of the 118 chemical elements that have been as of 2023. Across the second period Li and Be have distinguishing electrons in the 2s subshell, and electrons are being added to the 2p subshell in the atoms from B to Ne. In the first period the distinguishing electrons for H and He are in the 1s subshell. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The first three horizontal rows or periods in the modern periodic table consist entirely of representative elements. The first chemical element is Actinium and the last is Zirconium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is alphabethically listed. This list contains the 118 elements of chemistry. Click on the column header to sort the table by that column or click on an element name to get detailed facts about the element. Separation and Concentration Purification RequestĬhemical elements alphabetically listed The elements of the periodic table sorted by name in an alphabetical list.Ĭlick on any element's name for further chemical properties, environmental data or health effects. Here's a list of all of the chemical elements of the periodic table ordered by increasing atomic number.Plant Inspection & Process Optimalisation.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
